Pablo Gamallo (IQTC/UB) — DFT and kinetic study of methane dissociation over carbon catalysts for hydrogen production and carbon production
Don't miss any Success Story following us on X and LinkedIn!
@RES_HPC RES - Red Española de Supercomputación @res-icts.bsky.social
Check this Success Story at our LinkedIn: DFT and kinetic study of methane dissociation over carbon catalysts for hydrogen production and carbon production
💡Towards an industrial-scale clean hydrogen production 💡
📋"DFT and kinetic study of methane dissociation over carbon catalysts for hydrogen production and carbon production" led by Pablo Gamallo Belmonte from Institut de Química Teòrica i Computacional UB
Global hydrogen production mainly uses CO2-intensive processes, accounting for 2.5% of global CO2 emissions today. Methane pyrolysis is emerging as an attractive clean alternative that has no direct CO2 emissions, doesn't need water input and needs less energy than electrolysis. However, as it approaches industrial scale, catalyst design becomes increasingly critical. Carbon catalysts are abundant, inexpensive, and non-toxic, making them ideal for large-scale industrial use. In a methane pyrolisis reaction 3kg of solid carbon is produced per 1kg of hydrogen, and the structure of this carbon determines its value for applications (batteries, carbon fibers...)
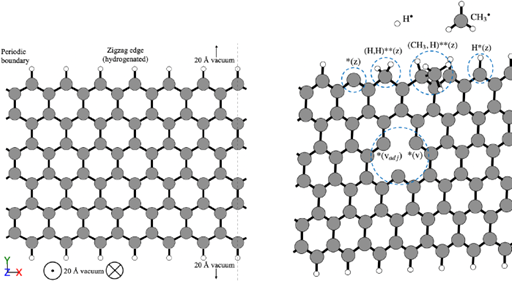
🖥️ Thanks to RES supercomputer #MareNostrum5 from Barcelona Supercomputing Center, the team examined methane, methyl, and hydrogen reactions with multiple surface sites (zig-zag, armchair edges, graphene vacancies, pentagonal rings, dangling aliphatic chains). They also calculated activation energies with the Nudged Elastic Band (NEB) method and modelled the reaction micro-kinetics, discovering that:
🔹 The most catalytically active site is a pentagonal ring originating from armchair sites.
🔹 Edge migration of hydrogen atoms is not energetically viable for prolonging catalyst activity.
🔹 Armchair edges are highly prone to pentagonal ring formation, leading to curvature and disorder in the carbon product.
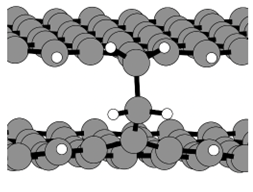
🔹 Extended aliphatic chains cause crosslinking between graphene sheets, creating defects that influence the material's suitability for battery applications.
📸 The images show (1) graphene flakes modelled in the simulations, fully saturated (left) and with presence of defects (right), (2) Cross-linking formation between graphene flakes
📋"DFT and kinetic study of methane dissociation over carbon catalysts for hydrogen production and carbon production" led by Pablo Gamallo Belmonte from Institut de Química Teòrica i Computacional UB
Global hydrogen production mainly uses CO2-intensive processes, accounting for 2.5% of global CO2 emissions today. Methane pyrolysis is emerging as an attractive clean alternative that has no direct CO2 emissions, doesn't need water input and needs less energy than electrolysis. However, as it approaches industrial scale, catalyst design becomes increasingly critical. Carbon catalysts are abundant, inexpensive, and non-toxic, making them ideal for large-scale industrial use. In a methane pyrolisis reaction 3kg of solid carbon is produced per 1kg of hydrogen, and the structure of this carbon determines its value for applications (batteries, carbon fibers...)
🖥️ Thanks to RES supercomputer #MareNostrum5 from Barcelona Supercomputing Center, the team examined methane, methyl, and hydrogen reactions with multiple surface sites (zig-zag, armchair edges, graphene vacancies, pentagonal rings, dangling aliphatic chains). They also calculated activation energies with the Nudged Elastic Band (NEB) method and modelled the reaction micro-kinetics, discovering that:
🔹 The most catalytically active site is a pentagonal ring originating from armchair sites.
🔹 Edge migration of hydrogen atoms is not energetically viable for prolonging catalyst activity.
🔹 Armchair edges are highly prone to pentagonal ring formation, leading to curvature and disorder in the carbon product.
🔹 Extended aliphatic chains cause crosslinking between graphene sheets, creating defects that influence the material's suitability for battery applications.
📸 The images show (1) graphene flakes modelled in the simulations, fully saturated (left) and with presence of defects (right), (2) Cross-linking formation between graphene flakes


